Sodium Bicarbonate: The Ubiquitous Alkali | Vibepedia
Sodium bicarbonate (NaHCO₃), commonly known as baking soda, is a white crystalline powder that's far more than a kitchen staple. Historically, its production…
Contents
- 🏠 Overview: The Kitchen Staple with a Chemical Soul
- 🔬 Chemical Identity & Origins
- 💡 Household Applications: Beyond the Baking Pan
- ⚕️ Health & Wellness Claims: Separating Fact from Fiction
- 🏭 Industrial Might: The Unsung Hero of Manufacturing
- ⚖️ Environmental Impact & Sustainability
- 🛒 Where to Find It: Accessibility and Forms
- 🤔 The Great Bicarb Debate: Purity and Potency
- Frequently Asked Questions
- Related Topics
Overview
Sodium bicarbonate (NaHCO₃), commonly known as baking soda, is a white crystalline powder that's far more than a kitchen staple. Historically, its production has evolved from mining natural deposits like trona to large-scale industrial synthesis, notably the Solvay process. Its alkaline properties make it a versatile agent in everything from leavening baked goods and neutralizing acids to industrial applications like flue gas desulfurization and fire suppression. While widely regarded as safe and beneficial in many contexts, its use in certain 'alternative' health remedies, particularly for cancer, remains highly controversial and lacks robust scientific backing, placing it on a spectrum of debated efficacy and potential harm.
🏠 Overview: The Kitchen Staple with a Chemical Soul
Sodium bicarbonate, or NaHCO3, is far more than just the leavening agent in your grandmother's cookies. This humble white powder, tasting faintly of salt and alkalinity, is a chemical workhorse with a history stretching back to ancient Egypt. While its most famous role is in the kitchen, its applications span from industrial cleaning to potential health remedies. Understanding its properties unlocks a world of practical uses, making it one of the most versatile compounds readily available to consumers and industries alike. Its ubiquity, however, often masks a complex interplay of sourcing, purity, and application.
🔬 Chemical Identity & Origins
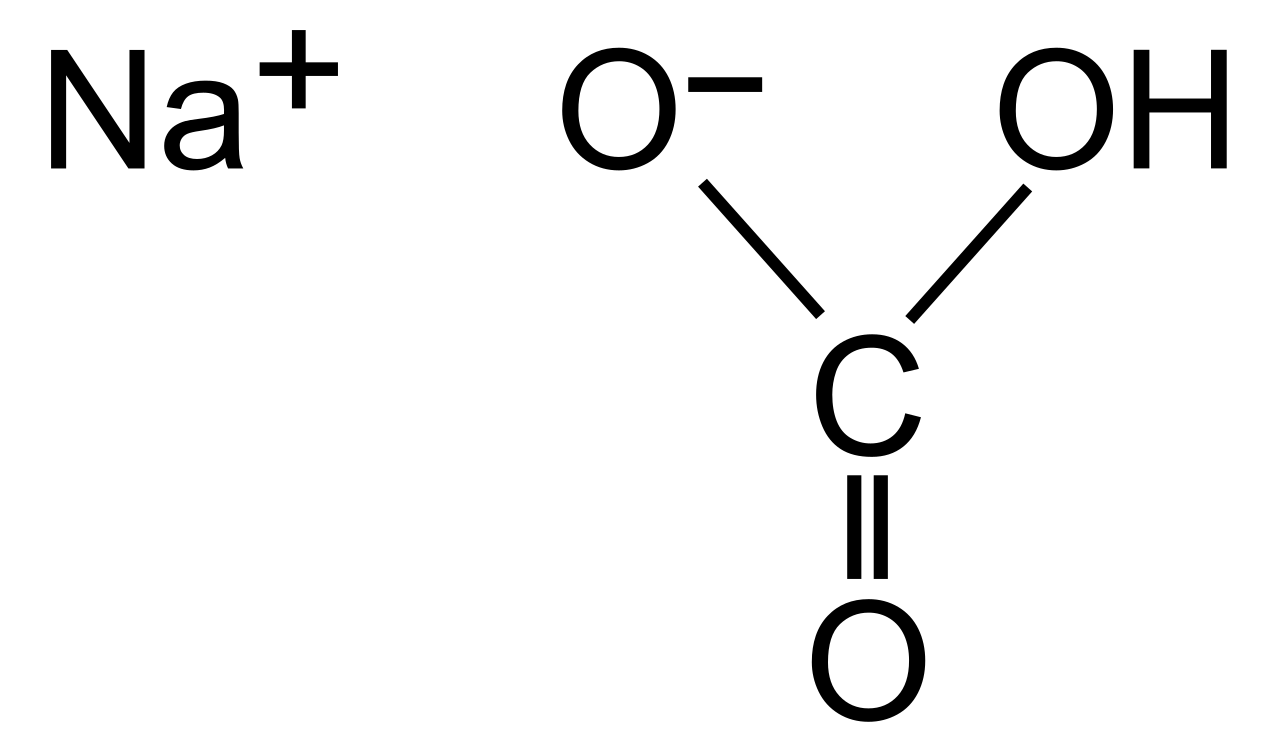
Chemically, sodium bicarbonate is a salt formed from a sodium cation (Na+) and a bicarbonate anion (HCO−3). Its IUPAC name is sodium hydrogen carbonate, and its natural mineral form is nahcolite, though it's often derived from the mineral trona. Historically, the production of sodium carbonate (soda ash) was a precursor, with the Solvay process, developed by Ernest Solvay in the 1860s, revolutionizing its industrial-scale synthesis. This process, which involves reacting brine with ammonia and carbon dioxide, made sodium bicarbonate far more accessible than earlier, more labor-intensive methods. The mineral trona, a sodium sesquicarbonate, is a significant natural source, particularly in regions like Green River, Wyoming.
💡 Household Applications: Beyond the Baking Pan
Beyond its culinary fame, sodium bicarbonate is a champion of the household. Its alkaline nature makes it an excellent deodorizer, absorbing odors in refrigerators and carpets alike. As a mild abrasive, it's a go-to for scrubbing sinks, countertops, and even tarnished silverware without scratching. Mixed with water, it forms a paste that can soothe insect bites and sunburns. Furthermore, it's a common ingredient in homemade cleaning solutions, often paired with vinegar for a fizzy, effervescent cleaning action that tackles grime and soap scum. Its versatility here is a testament to its pH buffering capabilities.
⚕️ Health & Wellness Claims: Separating Fact from Fiction
The health and wellness sphere buzzes with claims about sodium bicarbonate, from neutralizing stomach acid for heartburn relief to alkalinizing the body and even fighting cancer. While its antacid properties are well-established, with many over-the-counter remedies containing it, other claims remain controversial and largely unsubstantiated by rigorous scientific consensus. The idea of systemic alkalinization through dietary intake is particularly debated, with many medical professionals cautioning against self-treating serious conditions. The Vibepedia Controversy Spectrum for these health claims leans heavily towards 'Uncertain' to 'Disputed', demanding a critical approach to anecdotal evidence.
🏭 Industrial Might: The Unsung Hero of Manufacturing
Industrially, sodium bicarbonate is indispensable. It's used in fire extinguishers as a dry chemical agent, where it releases carbon dioxide upon heating, smothering flames. In the textile industry, it acts as a pH regulator and buffering agent. It's also crucial in leather tanning, wastewater treatment, and even in the production of certain pharmaceuticals and animal feed. The sheer volume of its industrial use, estimated in the millions of tons annually, underscores its importance as a fundamental chemical commodity, far exceeding its consumer-facing applications. Its role in flue gas desulfurization is also a significant, albeit less visible, contribution.
⚖️ Environmental Impact & Sustainability
From an environmental standpoint, sodium bicarbonate is generally considered safe and eco-friendly. Its natural mineral origins and relatively benign chemical properties mean it doesn't pose significant threats to ecosystems when used responsibly. Unlike harsher chemicals, it breaks down into readily available ions. However, the energy-intensive processes involved in its industrial production, particularly the Solvay process, do have an environmental footprint. Sustainable sourcing of trona and optimizing manufacturing efficiency are key areas for reducing its impact, aligning with broader goals of green chemistry.
🛒 Where to Find It: Accessibility and Forms
Sodium bicarbonate is remarkably accessible, found in virtually every supermarket's baking aisle under various names like 'baking soda' or 'bicarbonate of soda'. For industrial or higher-purity grades, it can be sourced from chemical suppliers. Consumer-grade baking soda typically has a purity of around 99%, suitable for most household and culinary uses. Pharmaceutical or food-grade versions, often labeled as such, offer higher purity standards for internal consumption or sensitive applications. The choice of grade depends entirely on the intended use, a crucial distinction for safety and efficacy.
🤔 The Great Bicarb Debate: Purity and Potency
A persistent debate surrounds the 'best' form of sodium bicarbonate, particularly concerning its use as an internal remedy. Some proponents advocate for specific brands or mineral sources, claiming superior efficacy or 'vibe'. Skeptics, however, point out that for most applications, the chemical compound NaHCO3 is the same, regardless of brand or origin, as long as it meets purity standards. The controversy often stems from anecdotal evidence and a misunderstanding of chemical equivalence versus perceived energetic properties. The Vibepedia Topic Intelligence on this subject highlights a tension between empirical chemical properties and subjective user experiences.
Key Facts
- Year
- 1846
- Origin
- United States (first commercial production)
- Category
- Chemical Compounds
- Type
- Chemical Compound
Frequently Asked Questions
Is baking soda the same as washing soda?
No, they are different. Baking soda is sodium bicarbonate (NaHCO3), while washing soda is sodium carbonate (Na2CO3). Sodium carbonate is a stronger alkali, more caustic, and primarily used for heavy-duty cleaning and laundry boosting, not for consumption or gentle scrubbing. Baking soda is milder and more versatile for household and culinary uses.
Can I use baking soda for heartburn relief?
Yes, sodium bicarbonate is a common ingredient in over-the-counter antacids and can provide rapid relief from heartburn by neutralizing stomach acid. However, it's important to use it as directed, as excessive consumption can lead to side effects like gas, bloating, and electrolyte imbalances. Consult a healthcare professional for persistent heartburn.
How does baking soda work as a deodorizer?
Sodium bicarbonate is amphoteric, meaning it can react with both acidic and basic odor molecules. It neutralizes them, effectively trapping and eliminating odors rather than just masking them. This makes it highly effective for absorbing smells in refrigerators, carpets, and pet areas.
What is the difference between food-grade and pharmaceutical-grade baking soda?
Both are highly purified forms of sodium bicarbonate. Food-grade is suitable for baking and general consumption. Pharmaceutical-grade meets even stricter purity standards set by regulatory bodies like the USP (United States Pharmacopeia) and is used in medicinal preparations and intravenous solutions, ensuring the highest level of safety and consistency.
Is sodium bicarbonate safe for the environment?
Generally, yes. Sodium bicarbonate is a naturally occurring compound and is considered environmentally benign. It breaks down into sodium and bicarbonate ions, which are common in natural water systems. The primary environmental concern relates to the energy-intensive production processes, not the compound itself once released.
Can baking soda be used to clean electronics?
It is generally not recommended to use baking soda to clean electronics. While it's a mild abrasive, the fine powder can potentially get into sensitive components and cause damage. Furthermore, any moisture used with baking soda could lead to short circuits. Specialized electronic cleaning solutions are safer and more effective.