Trophoblast Cells | Vibepedia
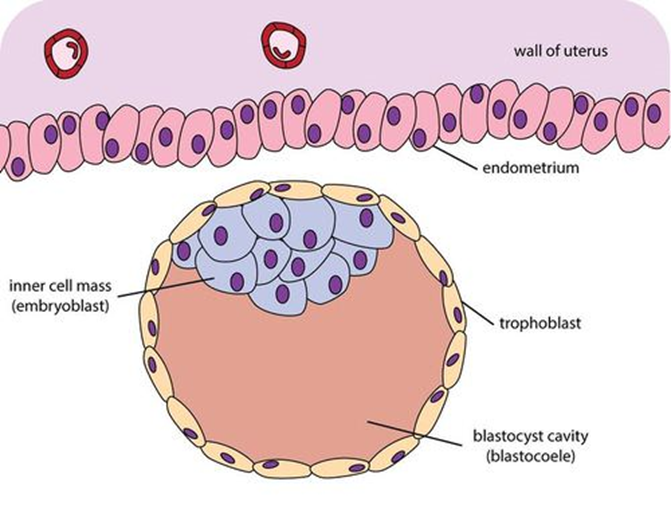
After initial differentiation, remaining cells in the human embryo transition from totipotency to pluripotency. The trophoblast's role is paramount…
Contents
Overview
The concept of trophoblast cells traces its origins to the earliest days of embryology, with foundational observations made in the late 19th and early 20th centuries as scientists began to unravel the complexities of mammalian reproduction. The term itself, derived from Greek 'trophein' (to feed) and 'blastos' (germinator), aptly describes their primary function. Walther Flemming and Oscar Hertwig laid groundwork in cell division and early embryonic development, paving the way for later identification of distinct cell lineages. The understanding of its role in immune tolerance and nutrient transfer has significantly evolved since the pioneering work of George W. Rüdi and others in the latter half of the 20th century.
⚙️ How It Works
Trophoblast cells function as the primary interface between the developing embryo and the maternal uterine environment. Upon reaching the blastocyst stage, typically around day 4 post-fertilization, the outer layer of cells differentiates into the trophoblast. These cells then invade the uterine lining (endometrium) to establish implantation. Subsequently, the trophoblast proliferates and differentiates into two main layers: the cytotrophoblast (inner layer of progenitor cells) and the syncytiotrophoblast (outer, multinucleated layer formed by fusion of cytotrophoblast cells). The syncytiotrophoblast is directly responsible for nutrient and gas exchange via the developing placenta, and it also produces critical hormones like human chorionic gonadotropin (hCG) that maintain the corpus luteum and pregnancy. Furthermore, trophoblast cells play a vital role in immune evasion, preventing the maternal immune system from rejecting the semi-allogeneic embryo.
📊 Key Facts & Numbers
Trophoblast cells are present as early as 4 days post-fertilization in humans. The placenta, which is largely derived from trophoblast cells, can weigh approximately 500 grams by the end of a full-term pregnancy. Trophoblast invasion into the uterine spiral arteries occurs between weeks 8 and 18 of gestation, a process critical for adequate blood supply. Studies indicate that approximately 1 in 10 pregnancies may experience complications related to placental development, often linked to trophoblast dysfunction. Growth Differentiation Factor 15 (GDF-15) is a protein produced by trophoblasts. Concentration of Growth Differentiation Factor 15 (GDF-15) has been correlated with the severity of nausea and vomiting in pregnancy (NVP), with levels potentially reaching several thousand picograms per milliliter in severe cases of hyperemesis gravidarum.
👥 Key People & Organizations
Key figures in understanding trophoblast biology include Robert Edward Man, whose early work on implantation was foundational, and Charles H. Baker, who contributed significantly to understanding placental development. The American Society for Reproductive Medicine and the Society for Reproductive Investigation are central hubs for research and dissemination of findings related to trophoblast biology and early pregnancy. Pharmaceutical companies such as BillionToOne are developing diagnostic tools that leverage understanding of placental markers, indirectly related to trophoblast function. Research institutions like Stanford University and Cambridge University host leading labs investigating trophoblast stem cells and their therapeutic potential.
🌍 Cultural Impact & Influence
The profound biological role of trophoblast cells has had a significant, albeit often indirect, cultural impact. The very concept of pregnancy and its successful continuation hinges on their function, influencing societal views on fertility, maternal health, and the miracle of life. Medical advancements stemming from trophoblast research, such as non-invasive prenatal testing (NIPT) which analyzes cell-free fetal DNA originating from trophoblasts, have revolutionized prenatal care and reduced the need for invasive procedures like chorionic villus sampling. The development of in vitro fertilization (IVF) technologies also relies heavily on understanding early embryonic development, including trophoblast formation. Cultural narratives often focus on the embryo and fetus, but the placenta, a trophoblast derivative, is increasingly recognized for its vital, complex role.
⚡ Current State & Latest Developments
Current research is intensely focused on understanding trophoblast stem cells (TSCs) and their potential for regenerative medicine and treating pregnancy disorders. Recent developments include the successful derivation and maintenance of human TSC lines, offering unprecedented opportunities to study early placental development and disease. For instance, studies in 2020-2023 have explored how viruses like SARS-CoV-2 interact with placental cells, revealing that while the virus rarely reaches first-trimester placentas, it can still disrupt early pregnancy immunity. Furthermore, breakthroughs in identifying the root causes of preeclampsia are emerging, with researchers investigating how to correct placental dysfunction, a condition directly linked to trophoblast abnormalities. The launch of diagnostic tests like BillionToOne's Unity Confirm™ highlights the growing clinical relevance of understanding placental biomarkers.
🤔 Controversies & Debates
A significant debate revolves around the precise origin and plasticity of trophoblast stem cells, particularly concerning their potential to differentiate into non-placental cell types. While widely accepted as the primary source of placental tissue, questions persist about their exact lineage and whether they can contribute to other embryonic tissues under specific conditions. Another area of contention is the role of maternal immune cells in trophoblast invasion and placental development; while immune tolerance is crucial, the exact mechanisms by which trophoblasts modulate the maternal immune response remain a subject of active investigation. The interpretation of abnormal trophoblast markers in non-invasive prenatal testing also presents ongoing challenges, with debates on sensitivity, specificity, and the clinical implications of borderline results.
🔮 Future Outlook & Predictions
The future of trophoblast research is exceptionally promising, particularly in the realms of reproductive health and regenerative medicine. Scientists are exploring the therapeutic potential of trophoblast stem cells for treating conditions beyond pregnancy complications, such as infertility and certain types of cancer, given their unique invasive and immune-modulating properties. Advances in gene editing technologies like CRISPR-Cas9 are expected to enable precise correction of genetic defects within trophoblast precursors, potentially preventing inherited diseases. Furthermore, the development of more sophisticated in vitro models of placental development, recapitulating the complex trophoblast-uterine interactions, will accelerate drug discovery and personalized treatment strategies for pregnancy disorders like preeclampsia and fetal growth restriction. The goal is to move from understanding to intervention, ensuring healthier pregnancies and potentially novel therapeutic avenues.
💡 Practical Applications
The most direct practical application of understanding trophoblast cells lies in prenatal diagnostics and the management of pregnancy complications. Non-invasive prenatal testing (NIPT), which analyzes cell-free DNA fragments shed by trophoblasts into the maternal bloodstream, is now a standard screening tool for chromosomal abnormalities like Down syndrome and Trisomy 18. Research into trophoblast markers is also crucial for early detection and management of conditions such as preeclampsia, gestational diabetes, and fetal growth restriction. Beyond pregnancy, the unique properties of trophoblast stem cells are being investigated for potentia
Key Facts
- Category
- science
- Type
- topic